ElectrochemistryHard
Question
The chemical reaction,
2AgCl(s) + H2(g) → 2HCl(aq) = 2Ag(s)
Taking place in a galvanic cell is represented by the notation :
2AgCl(s) + H2(g) → 2HCl(aq) = 2Ag(s)
Taking place in a galvanic cell is represented by the notation :
Options
A.Pt(s)|H2(g)′ 1 bar|1MKCl(aq)|AgCl(s)|Ag(g)
B.Pt(s)|H2(g)′ 1 bar|1MHCl(aq)|1MAg+(aq)|Ag(g)
C.Pt(s)|H2(g)′ 1 bar|1MHCl(aq)|AgCl(s)|Ag(g)
D.Pt(s)|H2(g)′ 1 bar|1MHCl(aq)|Ag(g)|AgCl(s)
Solution
In the given reaction 2AgCl(s) + H2(g)(1Bar) 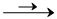 2HCl(aq) + 2Ag(s)
2HCl(aq) + 2Ag(s)
Silver is undergoing reduction(Ag+ → Ag)
hence it will act as cathode in the following cell.
Pt(s)|H2(g)1 bar|1MHCl(aq)|1MAg+(aq)|Ag(g)
(anode) (cathode)
(i) Thus, since option (a) has KCl which is not present in the cell, it is incrrect.
(ii) Since option (c) has AgCl(s) which does not ionise, it is incorrect,
(iii)Since in option(d),at cathode, Ag is being oxidised to Ag+ which is not possitive, so it is also incorrect.
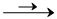 2HCl(aq) + 2Ag(s)
2HCl(aq) + 2Ag(s) Silver is undergoing reduction(Ag+ → Ag)
hence it will act as cathode in the following cell.
Pt(s)|H2(g)1 bar|1MHCl(aq)|1MAg+(aq)|Ag(g)
(anode) (cathode)
(i) Thus, since option (a) has KCl which is not present in the cell, it is incrrect.
(ii) Since option (c) has AgCl(s) which does not ionise, it is incorrect,
(iii)Since in option(d),at cathode, Ag is being oxidised to Ag+ which is not possitive, so it is also incorrect.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The standard electrode potentials (reduction) of Pt/Fe2+, Fe3+ and Pt/Sn4+, Sn2+ are + 0.77 V and 0.15 V respectively at...The highest electrical conductivity of the following aqueous solutions is of...The weight of silver (at. wt. = 108) displaced by a quantity of electricity which displaces 5600 mL of O2 at STP will be...Rust is a mixture of...If the pressure of hydrogen gas is increased from 1 atm. to 100 atm, keeping the hydrogen ion concentration constant at ...