ElectrochemistryHard
Question
The Gibbs energy for the decomposition of Al2O3 at 500oC is as follows :
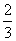 Al2O3 →
Al2O3 → 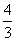 Al + O2, ᐃrG = + 966 kJ mol-1
Al + O2, ᐃrG = + 966 kJ mol-1
The potential difference needed for electrolytic reduction of Al2O3 at 500oC is at least
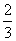 Al2O3 →
Al2O3 → 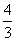 Al + O2, ᐃrG = + 966 kJ mol-1
Al + O2, ᐃrG = + 966 kJ mol-1The potential difference needed for electrolytic reduction of Al2O3 at 500oC is at least
Options
A.4.5 V
B.3.0 V
C.2.5 V
D.5.0 V
Solution
ᐃG = - nFE ⇒ E = 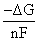
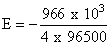
= - 2.5 V
∴ The potential difference needed for the reduction = 2.5 V
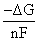
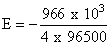
= - 2.5 V
∴ The potential difference needed for the reduction = 2.5 V
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Consider the cell Ag. The measured EMF of the cell is 1.0 V. What is the value of x ? EoAg+,Ag = + 0.8 V. [T = 25oC]...Mark out the correct statement(s)...A current of 2.0 ampere passed for 5 hours through a molten tin salt deposits 22.2 gm of metal. The oxidation number of ...The oxidation potentials of Zn, Cu, Ag, H2 and Ni are 0.76, -0.34, -0.80, 0.00, 0.25 volt, respectively. Which of the fo...The cell Zn|Zn²⁺(1M)||Cu²⁺(1M)|Cu with $E^\circ_{\text{cell}} = 1.10$ V was allowed to be completely discharged at 298 K...