ElectrochemistryHard
Question
In which of the following cells, EMF is greater than E-cell ?
Options
A.Pt, H2(g) | H+(pH = 5) || H+(pH = 3) | H2(g), Pt
B.Zn(s) | Zn2+(0.2 M) || Cu2+(0.1 M) | Cu(s)
C.Cr(s) | Cr3+(0.1 M) || Cu2+(0.2 M) | Cu(s)
D.Pt, H2(g) | H+ (pH = 4) || H+(pH = 6) | H2(g) | Pt
Solution
Use: Ecell = E-cell - 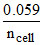 log Qcell
log Qcell
For Ecell > E-cell, Qcell < 1 (i.e., log Qcell < 0)
a. H2(1 atm) + 2H+ (10-3 M) → 2H+ (10-5 M) + H2 (1 atm)
Qcell =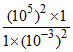 = 10 - 4 < 1
= 10 - 4 < 1
b . Zn(s) + Cu2+(0.1 M) → Zn2+ (0.2 M) + Cu(s)
Qcell =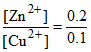 = 2 > 1
= 2 > 1
c. 2Cr(s) + 3Cu2+(0.2 M) → 2Cr3+ (0.1 M) + 3Cu(s)
Qcell =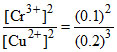 > 1
> 1
d. H2(1 atm) + 2H+ (10-6 M) → 2H+(10-4 M) + H2 (1 atm)
Qcell =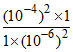 = 104 > 1
= 104 > 1
For Ecell > E-cell, Qcell < 1 (i.e., log Qcell < 0)
a. H2(1 atm) + 2H+ (10-3 M) → 2H+ (10-5 M) + H2 (1 atm)
Qcell =
b . Zn(s) + Cu2+(0.1 M) → Zn2+ (0.2 M) + Cu(s)
Qcell =
c. 2Cr(s) + 3Cu2+(0.2 M) → 2Cr3+ (0.1 M) + 3Cu(s)
Qcell =
d. H2(1 atm) + 2H+ (10-6 M) → 2H+(10-4 M) + H2 (1 atm)
Qcell =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which of the following facts is not true ?...Element A (atomic mass = 112) and element B (atomic mass = 27) form chlorides. Solutions of these chlorides are electrol...The oxidation states of sulphide in S8, S2F2, H2S respectively, are...For Na+, the value of symbol λm° is 50.0 Ω−1 cm2 mol−1. The speed of Na+ ion in the solution, if in the cell, electrodes...Calculate the solubility product of Co2[Fe(CN)6] in water at 25oC.Given, conductivity of saturated solutions of Co2[Fe(C...