ElectrochemistryHard
Question
Pt 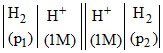 (where p1 and p2 are pressures) cell reaction will be spontaneous if :
(where p1 and p2 are pressures) cell reaction will be spontaneous if :
Options
A.p1 = p2
B.p1 > p2
C.p2 > p1
D.p1 = 1 atm
Solution
H2(P1) → 2H+ + 2e-
2H+ + 2e- → H2(P2)
H2(P2) → H2(P2)
E = 0 -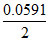 log
log 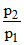
P2 < P1 for E → +ve
2H+ + 2e- → H2(P2)
H2(P2) → H2(P2)
E = 0 -
P2 < P1 for E → +ve
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which of the following statement(s) differentiate between electrochemical cell and electrolytic cell?...If the pressure of hydrogen gas is increased from 1 atm. to 100 atm, keeping the hydrogen ion concentration constant at ...Which of the following pair of metals, when coupled, will give maximum EMF for a voltaic cell?...Two platinum electrodes were immersed in a solution of CuSO4 and electric current was passed through the solution. After...On electrolysis, in which of the following, O2 would be liberated at the anode ?...