ElectrochemistryHard
Question
During the electrolysis of 0.1 M CuSO4 solution using copper electrodes, a depletion of [Cu++] occurs near the cathode with a corresponding excess near the anode, owing to inefficient stirring of the solution. If the local concentrations of [Cu++] near the anode & cathode are respectively 0.12 M & 0.08 M, calculate the back e.m.f. developed. Temperature = 298 K .
Options
A.22 mV
B.5.2 mV
C.29 mV
D.59 mV
Solution
Ecell = Eocell - 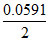 log10
log10 
Eocell = 0
Ecell = -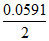 log10
log10 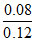 = + 5.2 mV.
= + 5.2 mV.
Eocell = 0
Ecell = -
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A current of 2.68 A is passed for one hour through an aqueous solution of CuSO4 using copper electrodes. Select the corr...The standard reduction potentials for Zn2+ / Zn,  Ni2+ / Ni and Fe2+ / Fe are - 0.76, - 0.23 and - 0.44 V respectiv...The equivalent conductances of two strong electrolytes at infinite dilution in H2O (where ions move freely through a sol...The standard EMF of a Daniel cell at 298 K is E1. When the concentration of ZnSO4 is 1.0 M and that of CuSO4 is 0.01 M, ...Given :E0(Cr+3/Cr) = -0.74 ; E0(MnO4-/Mn+2) = 1.51 VE0(Cr2O7-2/Cr+3) = 1.33 V; E0(Cl/Cl-) = 1.36 VBased on the data give...