ElectrochemistryHard
Question
The standard reduction potentials for Zn2+ / Zn, Ni2+ / Ni and Fe2+ / Fe are - 0.76, - 0.23 and - 0.44 V respectively. The reaction X + Y2+ → X2+ + Y will be spontaneous when
Options
A.X = Zn, Y = Ni
B.X = Ni, Y = Fe
C.X = Ni, Y = Zn
D.X = Fe, Y = Zn
Solution
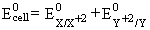
X = Zn Y = Ni
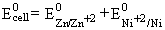
= 0.76 - 0.23 = 0.53 V
Eocell = + ve
ᐃGo = - ve so spontaneous process
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which of the following has longest C - O bond :...The electric charge for electrode deposition of one gram equivalent of a substance is :...Two students use same stock solution of ZnSO4 but different solutions of CuSO4. The EMF of one cell is 0.03 V higher tha...EMF of the cell: Cd(s)|CdCl2.5H2O (sat.)|AgCl(s)|Ag(s) is +0.70 V at 0°C and +0.60 V at 50°C. If ΔHo and ΔSo are tempera...In which of the following molecules π - electron density in ring is maximum :...