ElectrochemistryHard
Question
A current of 2.68 A is passed for one hour through an aqueous solution of CuSO4 using copper electrodes. Select the correct statement(s) from the following :
Options
A.increase in mass of cathode = 3.174 g
B.decrease in mass of anode = 3.174 g
C.no change in masses of electrodes
D.the ratio between the change of masses of cathode and anode is 1 : 2.
Solution
At Cathode :
Cu2+ + 2e- → Cu(s)
At Anode
Cu (s) → Cu2+ + 2e-
Increase in mass of cathode = decrease in mass of Anode =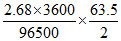 = 3.174 g.
= 3.174 g.
Cu2+ + 2e- → Cu(s)
At Anode
Cu (s) → Cu2+ + 2e-
Increase in mass of cathode = decrease in mass of Anode =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The specific conductance of a N/10 KCl at 25oC is 0.0112 ohm-1 cm-1. The resistance of cell containing solution at the s...A current of 2 A was passed for 1 h through a solution of CuSO4 0.237g of Cu2+ ions were discharged at cathode. The curr...100 mL of buffer of 1 M NH3(aq) and 1 M NH4+(aq) are placed in two comparments of a voltaic cell separately. A current o...The dissociation constant for CH3COOH is 1.8 × 10−5 at 298 K. The electrode potential for the half-cell:Pt|H2 (1 bar)|0....Which of the following statements would be true about this compound :...