ElectrochemistryHard
Question
If same quantity of electricity is passed through three electrolytic cells containing FeSO4, Fe2(SO4)3 and Fe(NO3)3, then
Options
A.the amount of iron deposited in FeSO4 and Fe2(SO4)3 are equal
B.the amount of iron deposited in FeSO4 is 1.5 times of the amount of iron deposited in Fe(NO3)3.
C.the amount of iron deposited in Fe2(SO4)3 and Fe(NO3)3 are equal
D.the same amount of gas is evolved in all three cases at the anode.
Solution
Fe2+ + 2e- → Fe [in FeSO4] ; Fe3+ + 3e- → Fe [in Fe2 (SO4)3]
Fe3+ + 3e- Fe [in Fe (NO3)3]
Amount of Fe deposited in FeSO4 =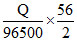
Amount of Fe deposited in Fe2(SO4)3 =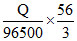
Fe3+ + 3e- Fe [in Fe (NO3)3]
Amount of Fe deposited in FeSO4 =
Amount of Fe deposited in Fe2(SO4)3 =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Standard electrode potential for Sn4+ / Sn2+ couple is +0.15 V and that for the Cr3+ / Cr couples is - 0.74. These two c...The value of molar conductivity of HCl is greater than that of NaCl at a particular temperature because...The EMFs of the cell obtained by combining separately Zn and Cu electrodes of a Daniel cell with normal calomel electrod...When iron is rusted, it is :...At 25oC molar conductance of 0.1 molar aqueous solution of ammonium hydroxide is 9.54 ohm-1 cm2mol-1 and at infinite dil...