ElectrochemistryHard
Question
When iron is rusted, it is :
Options
A.reduced
B.oxidised
C.evaporated
D.decomposed
Solution
Rusting reaction of Fe is -
2H+ + Fe +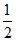 O2 → Fe2+ + H2O
O2 → Fe2+ + H2O
2H+ + Fe +
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The weight of silver (at. wt. = 108) displaced by a quantity of electricity which displaces 5600 mL of O2 at STP will be...During discharge of a lead storage cell the density of sulphuric acid in the cell :...Pt | Cl2 (P1 atm) | HCl (0.1 M) | Cl2 (P2 atm) | Pt, cell reaction will be spontaneous if...Any redox reaction would occur spontaneously, if :...The equivalent conductances of two strong electrolytes at infinite dilution in H2O (where ions move freely through a sol...