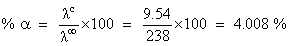ElectrochemistryHard
Question
At 25oC molar conductance of 0.1 molar aqueous solution of ammonium hydroxide is 9.54 ohm-1 cm2mol-1 and at infinite dilution its molar conductance is 238 ohm-1 cm2 mol-1. The degree of ionization of ammonium hydroxide at the same concentration and temperature is :-
Options
A.40.800%
B.2.080%
C.20.800%
D.4.008%
More Electrochemistry Questions
The two aqueous solutions; A (AgNO3) and B(LiCl) were electrolysed using Pt. electrodes. The pH of the resulting solutio...For the process: Cu2+ + 2e− → Cu; log[Cu2+] vs. Ered graph is shown in the figure, where OA = 0.34 V. The electrode pote...A hydrogen gas electrode is made by dipping platinum wire in a solution of HCl of pH = 10 and by passing hydrogen gas ar...For reducing one mole of Cr2O72- to Cr3+ the cahrge required is :...Which of the following solutions have highest resistance?...