ElectrochemistryHard
Question
The standard reduction potential of a silver chloride electrode is 0.2 V and that of a silver electrode is 0.79 V. The maximum amount of AgCl that can dissolve in 106 L of a 0.1 M AgNO3 solution is
Options
A.0.5 mmol
B.1.0 mmol
C.2.0 mmol
D.2.5 mmol
Solution
AgCl + e- → Ag + Cl- Eo = 0.2 V
Ag → Ag+ + e- Eo = - 0.79 V
_________________________________
AgCl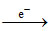 Ag+ + Cl- Eo = - 0.59 V
Ag+ + Cl- Eo = - 0.59 V
Eo =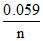 log K ⇒ - 0.59 =
log K ⇒ - 0.59 =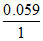 log KSP ⇒ KSP = 10-10
log KSP ⇒ KSP = 10-10
Now solubility of AgCl in 0.1 M AgNO3
S (S + 0.1) = 10-10 ⇒ S = 10-9 mol/L
Hence 1 mole dissolves in 109 L solution
hence in 106 L amount that dissolves in 1 m mol.
Ag → Ag+ + e- Eo = - 0.79 V
_________________________________
AgCl
Eo =
Now solubility of AgCl in 0.1 M AgNO3
S (S + 0.1) = 10-10 ⇒ S = 10-9 mol/L
Hence 1 mole dissolves in 109 L solution
hence in 106 L amount that dissolves in 1 m mol.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
An aqueous solution of X is added slowly to an aqueous solution of Y as shown in List-I. The variation in conductivity o...Given below are the half-cell reactions Mn2+ + 2e- → Mn ; Eo = - 1.18 V (Mn3+ + e- → Mn2+) ; Eo = + 1.51 V T...Rust is a mixture of...Which of the following statements is true for an electrochemical cell of Cu-H2....Choose the correct statement(s)....