ElectrochemistryHard
Question
Pt | Cl2 (P1 atm) | HCl (0.1 M) | Cl2 (P2 atm) | Pt, cell reaction will be spontaneous if
Options
A.P1 = P2
B.P1 > P2
C.P2 > P1
D.P1 = P2 = 1 atm
Solution
Ecell = Eocell - 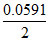 log10
log10 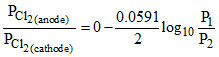
If P1 < P2, Ecell = + ve (spontaneous)
If P1 < P2, Ecell = + ve (spontaneous)
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Four colourless salt solutions are placed in separate test tubes and a strip of copper is placed in each. Which solution...Given standard electrode potentials : Fe3+ + 3e- → Fe ; Eo = - 0.036 voltFe2+ + 2e- → Fe; Eo = - 0.440 volt ...The current of 9.65 A flowing for 10 min deposits 3.0 g of a metal. The equivalent weight of the metal is...At equimolar concentrations of Fe2+ and Fe3+, what must [Ag+] be so that the voltage of the galvanic cell made from the ...The EMFs of the cell obtained by combining separately Zn and Cu electrodes of a Daniel cell with normal calomel electrod...