AIPMT | 2014ElectrochemistryHard
Question
The weight of silver (at. wt. = 108) displaced by a quantity of electricity which displaces 5600 mL of O2 at STP will be
Options
A.5.4 g
B.10.8 g
C.54.0 g
D.108.0 g
Solution
WO2 = 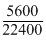 × 32 = 8 g = 1 equivalents
× 32 = 8 g = 1 equivalents
= 1 equivalent of Ag
= 108 g
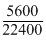 × 32 = 8 g = 1 equivalents
× 32 = 8 g = 1 equivalents= 1 equivalent of Ag
= 108 g
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Passage of 96,500 coulomb of electricity liberates ________ L of O2 at 273°C and 2 atm during electrolysis....Which of the following statements is true for an electrochemical cell of Cu-H2....The specific conductance of AgCl solution in water was determined to be 1.8 × 10−6 Ω−1 cm−1 at 298 K. The molar conducta...The standard reduction potential values of three metallic cations, x, y, z, are 0.52, - 1.18V respectively. The order of...At 25°C, the solubility product of CuCl is 2.0 × 10−7 and $E_{Cl^{-}|CuCl|Cu}^{o}$ is 0.128 V. The value of $E_{Cu^{+}|C...