ElectrochemistryHard
Question
ᐃG is the available energy (energy produced) during the electrochemical reaction in galvanic cell which can be converted into useful work. In the light of second law of thermodynamics in the cell the change in electrode potential with temperature will be equal to
Options
A.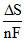
B.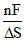
C.- 2.303 RT log KC
D.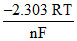
More Electrochemistry Questions
The ionization constant of a weak electrolyte is 25 × 10-6 while the equivalent conductance of its 0.01 M solution ...Both oxidation and reduction takes place in :...From the following E° values for the half-cells:(i) D → D2+ + 2e−; E° = −1.5 V (ii) B+ + e− → B; E° = − 0.5 V(iii) A3− →...A solution of sodium sulphate in water is electrolysed using inert electrodes. The products at the cathode and anode are...For the reduction of silver ions with copper metal, the standard cell potential was found to be +0.46 V at 25oC. The val...