ElectrochemistryHard
Question
The arrangement of X- ions around A+ ion in solid AX is given in the figure (not drawn to scale).If the radius of X- is 250 pm, the radius of A+ is -
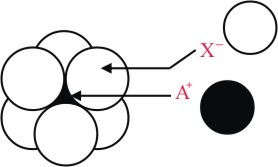

Options
A.104 pm
B.125 pm
C.183 pm
D.57 pm
Solution
Limiting radius ratio for octahedral void is 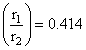
r+ = 0.414 × 250 pm ≃ 104 pm
Correct option (A)
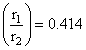
r+ = 0.414 × 250 pm ≃ 104 pm
Correct option (A)
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
What is the amount of chlorine evolved, when 2 amp of current is passed for 30 minutes in an aqueous solution of NaCl?...If Fe3+ + Y4- ⇋ FeY-; Kf = 1.3 × 1025Fe2+ + Y4- ⇋ FeY2-; Kf = 2.1 × 1014and Fe3+ + e- ⇋ Fe2+...Mark out the correct statement(s)...The standard reduction potentials E° of the following systems are System E° (volts)(i) MnO4- + 8H+ + 5e- → Mn2+ + ...The standard oxidation potentials, Eo, for the half reactions are asZn → Zn2+ + 2e- ; Eo = + 0.76 V → Fe ...