JEE AdvancedElectrochemistryHard
Question
Consider the electrode Ag | AgCl(s), Cl-(0.1 M), i.e., silver electrode in contact with 0.1 M KCl solution saturated with AgCl. If it is combined with the electrode Ag | Ag+(0.1 M) to from a complete cell, the EMF would be (Ksp od AgCl = 10-10 at 25oC)
Options
A.0.799
B.0.636
C.0.59
D.0.472
Solution
Ag | AgCl(s), Cl- (0.1 M) || Ag+ (0.1 M) | Ag
Ksp of AgCl = [Ag+] [Cl+]
10-10 = [Ag+] × 0.1
[Ag+]a = 10-9
For concentration cell, E- = 0
E = -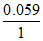 log
log 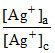
= - 0.059 log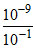
= - 0.059 × (-8) = 0.472
Ksp of AgCl = [Ag+] [Cl+]
10-10 = [Ag+] × 0.1
[Ag+]a = 10-9
For concentration cell, E- = 0
E = -
= - 0.059 log
= - 0.059 × (-8) = 0.472
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The oxidation states of iodine in HIO4, H3IO5 and H5IO6 are respectively...The correct order of EoM2+/M values with negative sign for the four successive elements Cr, Mn, Fe and Co is...The best conductor of electricity is a 0.1 M solution of...Faraday′s laws of electrolysis are related to the :...The standard reduction potential values of three metallic cations, X, Y and Z are +0.52, −3.03 and −1.18 V, respectively...