ElectrochemistryHard
Question
In H2 - O2 fuel cell the reaction occuring at cathode is :
Options
A. 2 H2O + O2 + 4 e- → 4OH-
B.2 H2 + O2 → 2 H2O (l)
C.H+ + OH- → H2O
D. H+ + e- → 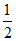 H2 .
H2 .
Solution
H2 - O2 fuel cell
At anode : 2OH- + H2 → 2H2O + 2e-
At cathode : 2 H2O + O2 + 4e- → 4OH-
At anode : 2OH- + H2 → 2H2O + 2e-
At cathode : 2 H2O + O2 + 4e- → 4OH-
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
These are three canonical structures of naphthalene. Examine them and find correct statement among the following :...In a galvanic cell...One g equivalent of Na metal is formed from electrolysis of fused NaCl. No. of mole of Al from the fused Na3AlF6 with th...With t taken in seconds and I taken in Amp, the variation of I follows the equation t2 + I2 = 25what amount of Ag will b...Calculate the solubility product of Co2[Fe(CN)6] in water at 25oC.Given, conductivity of saturated solutions of Co2[Fe(C...