ElectrochemistryHard
Question
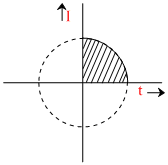
With t taken in seconds and I taken in Amp, the variation of I follows the equation
t2 + I2 = 25
what amount of Ag will be electrodeposited with this current flowing in the interval 0 - 5 second ? (Ag - 108)
t2 + I2 = 25
what amount of Ag will be electrodeposited with this current flowing in the interval 0 - 5 second ? (Ag - 108)
Options
A.22 mg
B.66 mg
C.77 mg
D.88 mg
Solution
Q =
∴ m =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The standard reduction potential values of three metallic cations, X, Y and Z are +0.52, −3.03 and −1.18 V, respectively...Element A (atomic mass = 112) and element B (atomic mass = 27) form chlorides. Solutions of these chlorides are electrol...Limiting molar conductivity of NH4OH (i.e. Λm (NH4OH)) is equal to :...Consider the reaction of extraction of gold from its ore:Au(s) + 2CN− (aq) + ¼ O2(g) + ½ H2O(l) → Au(CN)2−(aq) + OH−(aq)...Electrolysis of an acetate solution produces ethane according to the reaction:2CH3COO− → C2H6(g) + 2CO2(g) + 2e−What tot...