ElectrochemistryHard
Question
Calculate the solubility product of Co2[Fe(CN)6] in water at 25oC.
Given, conductivity of saturated solutions of Co2[Fe(CN)6] is 2.06 × 10-6 Ω-1 cm-1 and that of water used is 4.1 × 10-1 Ω-1 cm-1. The ionic molar conductivities of Co2+ and Fe(CN)64- are 86.0 Ω cm2 mol-1 and 444.0 Ω-1 cm2 mol-1, respectively.
Given, conductivity of saturated solutions of Co2[Fe(CN)6] is 2.06 × 10-6 Ω-1 cm-1 and that of water used is 4.1 × 10-1 Ω-1 cm-1. The ionic molar conductivities of Co2+ and Fe(CN)64- are 86.0 Ω cm2 mol-1 and 444.0 Ω-1 cm2 mol-1, respectively.
Options
A.7.87 × 10-7
B.7.87 × 10-6
C.7.87 × 10-8
D.7.87 × 10-9
Solution
KmCo2[F(CN)6 = 2.06 × 10-6 - 0.41 × 10-6 = 1.65 × 10-6
ΛmCo2[Fe(CN)6 = 2ΛmCo2+ + Λm[Fe(CN)6]4-
= 2 × 86 + 444 = 616 S cm2 mol-1
⇒ Λm = 1000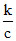 ⇒ c = 27 × 10-6 M
⇒ c = 27 × 10-6 M
KspCo2[Fe(CN)6] = 4c3 = 7.87 × 10-7 M3
ΛmCo2[Fe(CN)6 = 2ΛmCo2+ + Λm[Fe(CN)6]4-
= 2 × 86 + 444 = 616 S cm2 mol-1
⇒ Λm = 1000
KspCo2[Fe(CN)6] = 4c3 = 7.87 × 10-7 M3
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
C1 - C2 bond is shortest in...Which of the following rection is used to maker a fuel cell ?...A certain current liberated 0.504 g of hydrogen in 2 hours. How many grams of copper can be liberated by the same curren...The resistance of 0.1 N solution of formic acid is 200 ohm and cell constant is 2.0 cm-1. The equivalent conductivity (i...The correct order of EoM2+/M values with negative sign for the four successive elements Cr, Mn, Fe and Co is...