ElectrochemistryHard
Question
One g equivalent of Na metal is formed from electrolysis of fused NaCl. No. of mole of Al from the fused Na3AlF6 with the same current passed is :
Options
A.1
B.3
C.1/3
D.2
Solution
Na+ + e- → Na(s)
1mole 1 Faraday
Al3+ + 3e- → Al(s)
1 Faraday
No. of mole of Al =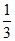 mole.
mole.
1mole 1 Faraday
Al3+ + 3e- → Al(s)
1 Faraday
No. of mole of Al =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A current of 9.95 amp following for 10 minutes, deposits 3 gm of a metal. Equivalent weight of the metal is...The oxidation number of sulphur in H2S2O7 is :...For the electrochemical cell, (M | M+) || (X- | X), Eo = (M+ | M) = 0.44V and Eo(X | X-) = 0.33V From this data one can ...How many gm of silver will be displaced from a solution of AgNO3 by 4 gm of magnesium?...Consider the following cell reaction:2Fe(s) + O2(g) + 4H+(aq) → 2Fe2+(aq) + 2H2O(l) Eo = 1.67V At [Fe2+] = 10-3 M,...