ElectrochemistryHard
Question
Using the data in the preceding problem, calculate the equilibrium constant of the reaction at 25oC.
Zn + Cu++ ⇋ Zn++ + Cu, K =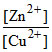
Zn + Cu++ ⇋ Zn++ + Cu, K =
Options
A.8.314 × 1024
B.4.831 × 1031
C.8.314 × 1036
D.4.831 × 1044
Solution
E•298 = 1.1028 - 0.641 × 10-3 × 25 + 0.72 × 10-5 × (25)2 = 1.091275 Volt
= - 8.314 × 298 ln K = - 2 × 1.091275 × 96500
K = 1036.91= 8.128 × 1036.
= - 8.314 × 298 ln K = - 2 × 1.091275 × 96500
K = 1036.91= 8.128 × 1036.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The electrical conductivity of a solution serves as a means of determining the end point in a chemical reaction, involve...What would be the magnitude of the EMF of the following cell:Pt(H2) =Pt(H2) at 25oC ? The ionization constant of acetic ...The standard reduction potentials E° of the following systems are System E° (volts)(i) MnO4- + 8H+ + 5e- → Mn2+ + ...In a H2 - O2 fuel cell, 6.72 L of hydrogen at NTP reacts in 15 minutes, the average current produced in amperes is...A current is passed through 2 voltameters connected in series. The first voltameter contains XSO4 (aq.) and second has Y...