ElectrochemistryHard
Question
What would be the magnitude of the EMF of the following cell:
Pt(H2) =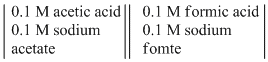 Pt(H2)
Pt(H2)
at 25oC ? The ionization constant of acetic acid, Ka ~ 10-5, while that of formic acid, Ka ~ 10-4.
Pt(H2) =
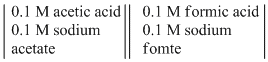 Pt(H2)
Pt(H2) at 25oC ? The ionization constant of acetic acid, Ka ~ 10-5, while that of formic acid, Ka ~ 10-4.
Options
A. 0.0295 V
B.0.059 V
C.-0.059 V
D.-0.0295 V
Solution
At anode:
0.1 M acetic acid + 0.1 M sodium acetate ⇒ Acidic buffer.
∴ pHa = pKa + log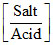 (pKa(CH3COOH) = 5)
(pKa(CH3COOH) = 5)
pHc = 4
Ecell = -0.059(pHc - pHa) = -0.059(4 - 5) = 0.059 V
0.1 M acetic acid + 0.1 M sodium acetate ⇒ Acidic buffer.
∴ pHa = pKa + log
pHc = 4
Ecell = -0.059(pHc - pHa) = -0.059(4 - 5) = 0.059 V
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Calculate the cell EMF in mV for Pt | H2 (1atm) | HCl (0.01 M) | AgCl(s) | Ag(s) at 298 Kif ᐃGfo values are at 25o...Given :E0(Cr+3/Cr) = -0.74 ; E0(MnO4-/Mn+2) = 1.51 VE0(Cr2O7-2/Cr+3) = 1.33 V; E0(Cl/Cl-) = 1.36 VBased on the data give...Chromium plating can involve the electrolysis of an electrolyte of an acidified mixture of chromic acid and chromium sul...Arrange following compounds in decreasing order of electrophilic substitution. (i) (ii) (iii) (iv)...Cost of electricity for the production of ′X′ litre H2 at NTP at cathode is Rs. X, then cost of electricity ...