ElectrochemistryHard
Question
The EMF of a concentration cell consisting of two zinc electrodes, one dipping into 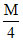 sol. of zinc sulphate & the other into
sol. of zinc sulphate & the other into 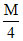 sol. of the same salt at 25oC is
sol. of the same salt at 25oC is
Options
A.0.0125 V
B.0.0250 V
C.0.0178 V
D.0.0356 V
Solution
E = 0 - 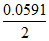 log
log 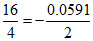 × 2 log 2 = - 0.0591 × 0.301 = - 0.0178 Volt.
× 2 log 2 = - 0.0591 × 0.301 = - 0.0178 Volt.
If connected in reverse direction, E = 0.0178 volt.
If connected in reverse direction, E = 0.0178 volt.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A current of 9.65 ampere is passed through the aqueous solution NaCl using suitable electrodes for 1000 s. The amount of...Which of the following molecules can act as an oxidizing as well as a reducing agent ?...The correct relationship between Gibb′s energy change in a reaction and the corresponding equilibrium constant KC ...The highest electrical conductivity of the following aqueous solutions is of...During electrolysis of water the volume of O2 Liberated is 2.24 dm3. The volume of hydrogen liberated, under same condit...