ElectrochemistryHard
Question
Which of the following molecules can act as an oxidizing as well as a reducing agent ?
Options
A.H2S
B.SO3
C.H2O2
D.F2
Solution
H2O2 can act both as an oxidising as well as a reducing agent.
Oxidising agent
pbS + 4H2O2 →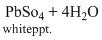
Reducing agent
Ag2O + H2O2 → 2Ag + H2O + O2
(moist)
Oxidising agent
pbS + 4H2O2 →
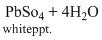
Reducing agent
Ag2O + H2O2 → 2Ag + H2O + O2
(moist)
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The specific conductance of a N/10 KCl at 25oC is 0.0112 ohm-1 cm-1. The resistance of cell containing solution at the s...Limiting molar conductivity of NH4OH (i.e. Λm (NH4OH)) is equal to :...The oxidation states of iodine in HIO4, H3IO5 and H5IO6 are respectively...The potential (EMF) of a cell consisting of an anode of silver in 0.10 M – AgNO3 solution and a cathode of Pt immersed i...In the lead storage battery, the anode reaction is Pb(s) + HSO4− + H2O → PbSO4(s) + H3O+ + 2e−. How many grams of Pb wil...