ElectrochemistryHard
Question
The standard potential of the reaction H2O + e- → 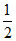 H2 + OH- at 298 K by using Kw (H2O) = 10-14, is :
H2 + OH- at 298 K by using Kw (H2O) = 10-14, is :
Options
A.- 0.828 V
B.0.828 V
C.0 V
D.- 0.5 V
Solution
H+ + e- → 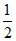 H2 , Eo = 0 ,ᐃGo = 0
H2 , Eo = 0 ,ᐃGo = 0
H2O ⇋ H+ + OH-, ᐃGo = - 8.314 × 298 ln 10-14
H2O + e- →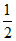 H2 + OH- , -1 × Eo × 96500 = - 8.314 × 298 ln10-14
H2 + OH- , -1 × Eo × 96500 = - 8.314 × 298 ln10-14
Eo = - 0.828 Volt.
H2O ⇋ H+ + OH-, ᐃGo = - 8.314 × 298 ln 10-14
H2O + e- →
Eo = - 0.828 Volt.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Any redox reaction would occur spontaneously, if :...A galvanic cell is composed of two hydrogen electrodes, one of which is a standard one. In which of the following soluti...The Gibbs energy for the decomposition of Al2O3 at 500oC is as follows : Al2O3 → Al + O2, ᐃrG = + 966 kJ mol...For a cell reaction involving two electron change, the standard EMF of the cell is 0.295 V at 2oC. The equilibrium const...Equivalent conductance of 1M CH3COOH is 10 ohm-1 cm2 equiv-1 and that at infinite dilution is 200 ohm–1 cm2, equiv-1. He...