ElectrochemistryHard
Question
A galvanic cell is composed of two hydrogen electrodes, one of which is a standard one. In which of the following solutions should the other electrode be immersed to get maximum emf ?
Ka (CH3COOH) = 2 × 10-5, Ka (H3PO4) = 10-3.
Ka (CH3COOH) = 2 × 10-5, Ka (H3PO4) = 10-3.
Options
A. 0.1 M HCl
B.0.1 M CH3COOH
C.0.1 M H3PO4
D.0.1 M H2SO4
Solution
Ecell = 0.059 log 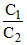
For Ecell to be +ve and maximum
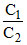 < 1 or C1 < C2 Give C2 = 1M.
< 1 or C1 < C2 Give C2 = 1M.
∴ C1 should be the minimum conc. of H+. ∴(B) is the right answer.
For Ecell to be +ve and maximum
∴ C1 should be the minimum conc. of H+. ∴(B) is the right answer.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
During an electrolysis of conc. H2SO4, perdisulphuric acid (H2S2O8) and O2 form in equimolar amount. The amount of H2 th...The standard EMF of a Daniel cell at 298 K is E1. When the concentration of ZnSO4 is 1.0 M and that of CuSO4 is 0.01 M, ...A current of 0.1A was passed for 2hr through a solution of cuprocyanide and 0.3745 g of copper was deposited on the cath...CH3O - CH = CH - NO2 ICH2 = CH - NO2 IICH2 = CH - Cl IIICH2 = CH2 IV Which of the following is the correct order of C - ...The following galvanic cell: Zn|Zn(NO3)2 (100 ml, 1 M)||Cu(NO3)2 (100 ml, 1 M)|Cu was operated as an electrolytic cell a...