ElectrochemistryHard
Question
For a cell reaction involving two electron change, the standard EMF of the cell is 0.295 V at 2oC. The equilibrium constant of the reaction at 25oC will be
Options
A.25.5 × 10-2
B.10
C.1 × 1010
D.2.95 × 10-10
Solution
Eocell = 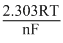 logKc =
logKc = 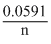 logKc
logKc
∴ 0.295V =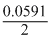 logKc
logKc
or log Kc =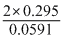 = 10
= 10
or Kc = 1 × 1010
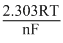 logKc =
logKc = 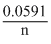 logKc
logKc∴ 0.295V =
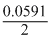 logKc
logKcor log Kc =
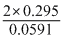 = 10
= 10or Kc = 1 × 1010
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
In the reaction I2 = I- → I3-, the Lewis base is :...An acidic solution of copper (II) sulphate containing some contaminations of zinc and iron (II) ions was electrolysed ti...When a lead storage battery is charged...At 25oC molar conductance of 0.1 molar aqueous solution of ammonium hydroxide is 9.54 ohm-1 cm2mol-1 and at infinite dil...In the lead storage battery, the anode reaction is Pb(s) + HSO4− + H2O → PbSO4(s) + H3O+ + 2e−. How many grams of Pb wil...