Chemical Kinetics and Nuclear ChemistryHard
Question
Rate constant K = 2.303 min-1 for a particular reaction. The initial concentration of the reaction is 1 mol/litre then rate of reaction after 1 minute is ?
Options
A.2.303 M min-1
B.0.2303 M min-1
C.0.1 M min-1
D.None of these
Solution
K = 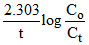
2.303 =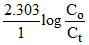
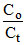 = 10
= 10
Ct = 0.1
Rate = K [Ct]
= 2.303 × 0.1 = 0.2303 M min-1
2.303 =
Ct = 0.1
Rate = K [Ct]
= 2.303 × 0.1 = 0.2303 M min-1
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Deep orange color of potasium dichromate is due to :-...The rate constant, the activation energy and the Arrhenious parameter of a chemical reaction at 25oC are 3.0 × 10-4...Number of Faraday required to liberate 8 g. of H2 is :-...A reaction of first-order completed 90% in 90 minutes, hence, it is completed 50% in approximately :...The rate law for a reaction between the substances A and B is given by Rate = k[A]n [B]m On doubling the concentration o...