Chemical Kinetics and Nuclear ChemistryHard
Question
The rate constant for the reaction, 2N2O5 → 4NO2 is 3.0 × 10-5s-1 If the rate is 3.40 × 10-5M sec-1, then the concentration of N2O5 (in M) is :
Options
A.1.4
B.1.2
C.0.04
D.0.8
Solution
Reaction is of I order as unit of rate constant is time-1(i.e.,sec-1),thus
rate = k[N2O5]
[N2O5] =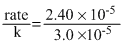 = 0.8M
= 0.8M
rate = k[N2O5]
[N2O5] =
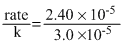 = 0.8M
= 0.8MCreate a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The order of a reaction is zero. It will be definitely...Decrease in atomic number is observed during :...For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...For the consecutive first-order reactions: $A\overset{\quad K_{1}\quad}{\rightarrow}B\overset{\quad K_{2}\quad}{\rightar...In the reaction : P + Q →R + S the time taken for 75% reaction of P is twice the time taken for 50% reaction of P. ...