ThermodynamicsHard
Question
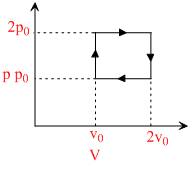
The above p-v diagram represents the thermodynamic cycle of an engine, operating with an ideal monoatomic gas. The amount of heat, extracted from the source in a single cycle is :
Options
A.p0v0
B.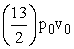
C.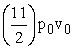
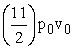
D.4p0v0
Solution
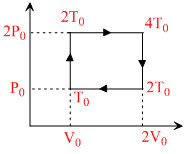
heat supplied = nCv(2T0 - T0) + nCp (4T0 - 2T0)
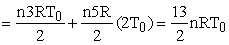
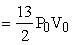
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
An ideal gas is taken around the cycle ABCA as shown in the P - V diagram. The net work done by the gas during the cycle...A diatomic ideal gas is heated under constant pressure, the part of heat that increase internal energy of gas is :-...Three closed vessels A, B and C at the same temperature T and contain gases which obey the Maxwellian distribution of ve...A gas is taken through the cycle A → B → C → A, as shown, What is the net work done by the gas ?...A system is taken along the paths A and B as shown. If the amounts of heat given in these processes are ᐃQA and &#...