ThermodynamicsHard
Question
Three closed vessels A, B and C at the same temperature T and contain gases which obey the Maxwellian distribution of velocities. Vessel A contain only O2, B only N2 and C a mixture of equal quantities of O2 and N2. If the average speed of the O2 molecules in vessel A is v1 that of the N2 molecules in vessels B is v2, the average speed of O2 molecules in vessel C is
Options
A.(v1 + v2) /2
B.v1
C.(v1 v2)1/2
D.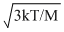
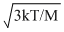
Solution
The average speed of molecules of an ideal gas is given by
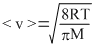 i.e., < v > ∝ √T for same gas
i.e., < v > ∝ √T for same gas
Since, temperature of A and C are same, average speed of O2 molecules will be equal in A and C i.e., v1∴ Correct option is (b).
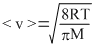 i.e., < v > ∝ √T for same gas
i.e., < v > ∝ √T for same gas Since, temperature of A and C are same, average speed of O2 molecules will be equal in A and C i.e., v1∴ Correct option is (b).
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Graphs between PV diagram for isothermal and adiabatic processes are drawn the relation between their slopes will be: -...In an adiabatic change, the pressure and temperature of a monoatomic gas are related as P ∝ TC, Where C equals...Show below are the black body radiation curves at temperature T1 and T2 (T2 > T1). Which of the following plots is corre...One mole ideal gas is compressed adiabatically at 27oC. Its temperature becomes 102oC. The work done in this process wil...The radiation from the sun, Incident normally at the surdace of the earth is 20 kcal/m2 min.What would have the radiant ...