SolutionHard
Question
Consider following terms (m = molality) :
I : mKb ; II : mKbi
III :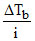 IV : Kb
IV : Kb
Terms which can be expressed in degree (temperature) are
I : mKb ; II : mKbi
III :
Terms which can be expressed in degree (temperature) are
Options
A.III, IV
B.I, II
C.I, II, IIII
D.I, III
Solution
As ᐃTb = i Kbm
so iKb m can be expressed in degree (Unit of temperature)
and Kb m can be expressed in degree (Unit of temperature)
and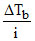 can be expressed in degree (Unit of temperature)
can be expressed in degree (Unit of temperature)
But unit of Kb is mol-1 kg K
so iKb m can be expressed in degree (Unit of temperature)
and Kb m can be expressed in degree (Unit of temperature)
and
But unit of Kb is mol-1 kg K
Create a free account to view solution
View Solution FreeMore Solution Questions
The relationship between osmotic pressure at 273 K when 10 g glucose (π1), 10 g urea (π2) and 10 g sucrose (π3) are diss...FeCl3 solution (side X) on reaction with K4[Fe(CN)6] (side Y) in aqueous solution gives blue colour. These are separated...20 g of a substance were dissolved in 500 mL of water and the osmotic pressure of the solution was found to be 600 mm of...The vapour pressure of water at room temperature is lowered by 5% on dissolving a non-volatile solute in it. The molalit...A complex containing K+, Pt(IV) and Cl- is 100% ionised giving i = 3. Thus, complex is :...