Atomic StructureHard
Question
A sodium street light gives off yellow light that has a wavelength of 600 nm. Then
(For energy of a photon take E =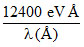 )
)
(For energy of a photon take E =
Options
A.frequency of this light is 7 × 1014 s-1
B.frequency of this light is 5 × 1014 s-1
C.wavenumber of the light is 3 × 106 s-1
D.energy of the photon is approximately 2.07 eV
More Atomic Structure Questions
The configuration 42H > 73Ne > 94 Be shows :...In a sample of H-atoms, electrons de-excite from a level ′n′ to 1. The total number of lines belonging to Ba...For the Hydrogen spectrum, last line of the Lyman series has frequency v1, last line of Lymen series of He+ ion has freq...What is the distance travelled by an electron revolving in the second orbit of Be3+ ion in 100 revolutions?...Some hydrogen-like atoms in ground state absorbs ‘n’ photons having same the energy and on de-excitement, it emits exact...