Atomic StructureHard
Question
For the Hydrogen spectrum, last line of the Lyman series has frequency v1, last line of Lymen series of He+ ion has frequency v2 and last line of Balmer series of He+ ion has frequency v3 then
Options
A.2(v1 + v3) = v2
B.v1 = v3
C.4v1 = v2
D.v2 = v3
Solution
v1 = Rc(1)2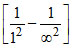 = Rc.
= Rc.
v2 = Rc(2)2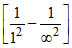 = 4Rc.
= 4Rc.
v3 = Rc(2)2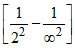 = Rc.
= Rc.
∴ 2(v1 + v3) = v2 and v1 = v3 and 4v1 = v2
v2 = Rc(2)2
v3 = Rc(2)2
∴ 2(v1 + v3) = v2 and v1 = v3 and 4v1 = v2
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The kinetic energy of an electron in the second Bohr orbit of a hydrogen atom is (ao is the Bohr radius)a) $\frac{h^{2}}...Ground state electronic configuration of nitrogen atom can be represented by :...The dynamic mass (in kg) of the photon with a wavelength corresponding to the series limit of the Balmer transitions of ...Bond dissociation on energy of Br2 is 200kJ/mole. The longest wavelength of photon that can break this bond would be (NA...The uncertainty in position and velocity of the particle are 0.1 nm and 5.27 × 10-27 ms-1 respectively then the mas...