Atomic StructureHard
Question
In a sample of H-atoms, electrons de-excite from a level ′n′ to 1. The total number of lines belonging to Balmer series are two. If the electrons are ionised from level ′n′ by photons of energy 13 eV. Then the kinetic energy of the ejected photoelectrons will be
Options
A.12.15 eV
B.11.49 eV
C.12.46 eV
D.12.63 eV
Solution
Number of lines in Balmer series = 2. ∴ n = 4 (lines will be 4 → 2, 3 → 2).
KE of ejected photoelectrons = Ephoton - BEn = 13 -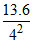 = 13 - 0.85 = 12.15 eV.
= 13 - 0.85 = 12.15 eV.
KE of ejected photoelectrons = Ephoton - BEn = 13 -
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The electron in the same orbital may be identified with the quantum number...The ionization energy of He+ ion is x eV. The ionization energy of Be3+ ion should be...An α-particle having kinetic energy 4.0 MeV is projected towards tin nucleus (Z = 50). Select the correct information(s)...The number of waves made by a Bohr electron in an orbit of maximum magnetic quantum number +2 is -...The species which has its fifth ionisation potential equal to 340 V is...