Atomic StructureHard
Question
An electron in a hydrogen like atom makes transition from a state in which its de-Broglie wavelength isλ1 to a state where its de-Broglie wavelength is λ2 then wavelength of photon (λ) generated will be
where m is mass of the electron, c is speed of light in vaccum.
where m is mass of the electron, c is speed of light in vaccum.
Options
A.λ = λ1 - λ2
B.λ = 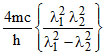
C.λ = 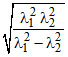
D.λ = 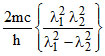
Solution
∴ λ =
∴
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following pairs of ions are isoelectronic and isostructural ?...Suppose that a hypothetical atom gives a red, green, blue and violet line spectrum. Which jump according to figure would...As the orbit number increases, the distance between two consecutive orbits (r1 = radius of first orbit)...The ratio of wavelengths of Kα-characteristic X-rays produced when iron (Z = 26) and scandium (Z = 21) are used as antic...An antiproton has the mass of a proton but a charge of –e. If a proton and an antiproton orbited each other, then how fa...