Atomic StructureHard
Question
Suppose that a hypothetical atom gives a red, green, blue and violet line spectrum. Which jump according to figure would give off the red spectral line.
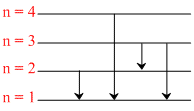

Options
A.3 → 1
B.2 → 1
C.4 → 1
D.3 → 2
Solution
According to energy, E4 → 1 > E3 → 1 > E2 → 1 > E3 → 2 .
According to energy, Violet > Blue > Green > Red.
∴ Red line ⇒ 3 → 2 transition.
According to energy, Violet > Blue > Green > Red.
∴ Red line ⇒ 3 → 2 transition.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The orbital angular momentum of a 4p electron will be...Uncertainty in the position of an electron (mass = 9.1 × 10−31 kg) moving with a velocity 300 m/s accurate up to 0.001% ...A completely filled d-orbital (d10) is of...Metal ion has d4 configuration in octahedral complex entity of low spin, the number of unpaired electron present in t2g ...The radionucleide undergoes two successive β - decays followed by one α - decay. The atomic numberand the mass...