Atomic StructureHard
Question
In a H-like sample, electrons make transition from 4th excited state to 2nd state then
Options
A.10 different spectral lines are observed
B.6 different spectral lines are observed
C.number of lines belonging to the blamer series is 3
D.Number of lines belonging to paschal series is 2.
Solution
Transition is taking place from 5 → 2
⇒ ᐃn = 3
Hence maximum number of spectral line observed =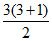
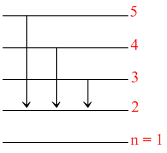
(C) number of lines belonging to the Balmer series = 3 (5 → 2,4 → 2,3→2) as shown in figure.
⇒ ᐃn = 3
Hence maximum number of spectral line observed =

(C) number of lines belonging to the Balmer series = 3 (5 → 2,4 → 2,3→2) as shown in figure.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following suggested that de Broglie wavelength is not possible for the electron in a Bohr's orbit of the hy...O2 undergoes photochemical dissociation into one normal oxygen atom and one oxygen atom, 1.2 eV more energetic than norm...The eyes of a certain member of the reptile family pass a single visual single to the brain when the visual receptors ar...An α-particle having kinetic energy 4.0 MeV is projected towards tin nucleus (Z = 50). Select the correct information(s)...The angular momentum of electron revolving in the second orbit of H-atom is ‘x’ J·s. The angular momentum of electron in...