Atomic StructureHard
Question
In a sample of H-atom electrons make transition from 5th excited state to ground state, producing all possible types of photons, then number of lines in infrared region are
Options
A.4
B.5
C.6
D.3
Solution
infrared lines = total lines - visible lines - UV lines = 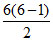 - 4 - 5 = 15 - 9 = 6.
- 4 - 5 = 15 - 9 = 6.
(visible lines = 4 6 → 2, 5 → 2, 4 → 2, 3 → 2) (UV lines = 5 6 → 1, 5 → 1, 4 → 1, 3 → 1, 2 → 1)
(visible lines = 4 6 → 2, 5 → 2, 4 → 2, 3 → 2) (UV lines = 5 6 → 1, 5 → 1, 4 → 1, 3 → 1, 2 → 1)
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The orbital angular momentum of an electron in 2s orbital is :...The uncertainty in position and velocity of the particle are 0.1 nm and 5.27 × 10-27 ms-1 respectively then the mas...The group in which all the elements do not have same number of electrons in their valence shell is :-...The orbital angular momentum for an electron revolving in an orbit is given by . This momentum for an s-electron will be...If there are only two H-atoms, each is in 3rd excited state then :...