Atomic StructureHard
Question
The orbital angular momentum for an electron revolving in an orbit is given by 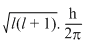 . This momentum for an s-electron will be given by
. This momentum for an s-electron will be given by
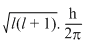 . This momentum for an s-electron will be given by
. This momentum for an s-electron will be given byOptions
A.zero
B.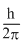
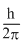
C.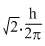
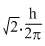
D.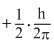
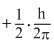
Solution
For s-electron, l = 0 ∴ angular momentum = zero
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The first use of quantum theory to explain the structure of atom was made by :...The value of Planck′s constant is 6.63 × 10-34 Js. The velocity of light is 3 × 108 m/sec. Which value i...The number of unpaired electron in G. S., first E.S. and second E.S. of S (Z = 16) are, respectively,...Which of the following has the maximum number of unpaired electrons ?...Positron is :...