Atomic StructureHard
Question
Which is the correct relationship :
Options
A.E1 of H = 1/2 E2 of He+ = 1/3 E3 of Li2+ = 1/4 E4 of Be3+
B.E1(H) = E2(He+) = E3 (Li2+) = E4(Be3+)
C.E1(H) = 2E2(He+) = 3E3 (Li2+) = 4E4(Be3+)
D.No relation
Solution
E1 (H) = - 13.6 × 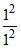 = - 13.6 eV ; E2 (He+) = - 13.6 ×
= - 13.6 eV ; E2 (He+) = - 13.6 × 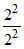 = - 13.6 eV
= - 13.6 eV
E3 (Li2+) = - 13.6 ×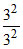 = - 13.6 eV ; E4 (Be3+) = - 13.6 ×
= - 13.6 eV ; E4 (Be3+) = - 13.6 × 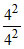 = - 13.6 eV
= - 13.6 eV
∴ E1(H) = E2(He+) = E3 (Li2+) = E4(Be3+)
E3 (Li2+) = - 13.6 ×
∴ E1(H) = E2(He+) = E3 (Li2+) = E4(Be3+)
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A sodium street light gives off yellow light that has a wavelength of 600 nm. Then (For energy of a photon take E = )...Identify the correct statement(s) :...A gas absorbs a photon of 355 nm and emits at two wavelengths. If one of the emissions is at 680 nm, the other is at :...Incorrect statement is :-...Biotite has the emperical formula of (Si2O5)x2x- . Find the number of oxygen atom shared per tetrahedron...