Atomic StructureHard
Question
Identify the correct statement(s) :
Options
A.Wavelength associated with the 1 kg ball moving with the velocity 100 m/s can′t be calculated.
B.Wave nature of the running train is difficult to observe because wavelength is extremely small.
C.Wavelength associated with the electron can be calculated using the formulae E = 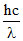
D.If an electron is accelerated through 20 V potential difference if it has already 5eV kinetic energy then wavelength of the electron is approximately √6 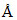 .
.
Solution
(A) λ can be calculated as : λ = 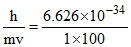 = 6.626 × 10-36 m. (very small).
= 6.626 × 10-36 m. (very small).
(B) de-Broglie wavelength associated with macroscopic particles is extremely small and so, difficult to observe.
(c) de-Broglie wavelength associated with electron can be calculated by using λ =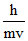 .
.
(D) KEf = 5 + 20 = 25 eV.
∴ λ =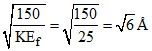
(B) de-Broglie wavelength associated with macroscopic particles is extremely small and so, difficult to observe.
(c) de-Broglie wavelength associated with electron can be calculated by using λ =
(D) KEf = 5 + 20 = 25 eV.
∴ λ =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following compounds, conjugate base is stablized by d-orbital resonance -...There are two samples of H and He atom. Both are in some excited state. In hydrogen atom total number of lines observed ...When an excited hydrogen atom returned to its ground state, some visible quanta were observed along with other quanta. W...The magnitude of the spin angular momentum of an electron is given by...The increasing order (lowest first) for the values of e/m (charge /mass) for electron (p),neutron (n) and alpha particle...