Solid StateHard
Question
An ionic solid of XY type having anions in CCP lattice and cations in the octahedral voids.Let a be the edge length of an FCC cube.The radius ratio of cation(R+) to that of anion(R-)is greater than 0.415.Then which of the following is false:
Options
A.R+ =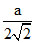
B.R+ + R- = 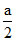
C.Anions will not be in contact with each other.
D.Cations will not be in contact with each other.
More Solid State Questions
The empty space between the shaded balls and hollow balls as shown in the diagram is called...Aluminum metal has a density of 2.72 g/cm3 and crystallizes in a lattice with an edge length of 404 pm.Which of the foll...Sodium metal crystallizes in a body centred cubic lattice with a unit cell edge of 4.29(Ao). The radius of sodium atom i...If the anions (A) form hexagonal closest packing and cations (C) occupy only 2/3 octahedral voids in it, then the genera...In a body centered cubic cell (bcc) of lattice parameter a, the atomic radius is :-...