Solid StateHard
Question
Sodium metal crystallizes in a body centred cubic lattice with a unit cell edge of 4.29(Ao). The radius of sodium atom is approximately :-
Options
A.5.72(Ao)
B.0.93(Ao)
C.1.86(Ao)
D.3.022(Ao)
Solution
For BCC, √3 × a = 4R
∴ R =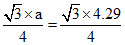 = 1.86 (Ao)
= 1.86 (Ao)
∴ R =
Create a free account to view solution
View Solution FreeMore Solid State Questions
The crystalline structure of a solid is CCP for ‘X’ atoms. If ‘Y’ atoms occupy all the octahedral voids and ‘Z’ atoms oc...An ionic solid is HCP of Q2− ions and Px+ ions are in half of the tetrahedral voids. The value of x should be...Na and Mg crystallize in BCC and FCC type crystals respectively, then the number of atoms of Na and Mg present in the un...In a sodium chloride structure, the percentage of the octahedral voids occupied by cations is :...CsBr has CsCl type structure. Its density is 4.26 g/cm³. Calculate the edge length of the unit cell. (Given: Cs = 133, B...