Solid StateHard
Question
In a body centered cubic cell (bcc) of lattice parameter a, the atomic radius is :-
Options
A.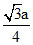
B.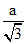
C.2√2a
D.a/4
More Solid State Questions
In a face centered cubic cell, the contribution of an atom at a face of the unit cell is :...The composition of a sample of cuprous oxide is found to be Cu1.92O1.00 due to metal deficient defect. The molar ratio o...MgO exists in a rock salt type unit cell. Each Mg2+ ion will be in contact with...Square packed sheets are arranged on the top of other such that a sphere in the next layer rests on the center of a squa...An element occurring in the body-centred cubic (BCC) structure has 1.208 × 1023 unit cells. The total number of atoms of...