Atomic StructureHard
Question
Which transition in Li2+ would have the same wavelength as the 2 → 4 transition in He+ ion ?
Options
A.4 → 2
B.2 → 4
C.3 → 6
D.6 → 2
Solution
(He+)2 → 4 = (Li2+)n4 → n3
∴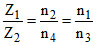 or
or 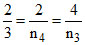
∴ n4 = 3 and n3 = 6.
∴ Transition in Li2+ ion = 3 → 6
∴
∴ n4 = 3 and n3 = 6.
∴ Transition in Li2+ ion = 3 → 6
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
If the value of En = - 78.4 kcal/mole, the order of the orbit in hydrogen atom is :...Cumbrous ion is collarless while cupric ion is colored because...In presence of HCl, metallic tin (Sn) is oxidised into stannic chloride with the help of K2Cr2O7. How many mL of 0.1 N K...When KMnO4 acts as an oxidising agent and ultimately forms [MnO4]-1, MnO2, Mn2O3, Mn+2 then the number of electrons tran...Many elements have non-integral atomic masses because...