Atomic StructureHard
Question
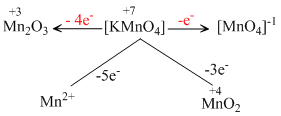
When KMnO4 acts as an oxidising agent and ultimately forms [MnO4]-1, MnO2, Mn2O3, Mn+2 then the number of electrons transferred in each case respectively is
Options
A.4, 3, 1, 5
B.1, 5, 3, 7
C.1, 3, 4, 5
D.3, 5, 7, 1
More Atomic Structure Questions
Which of the following statements is/are correct for an electron of quantum numbers n = 4 and m = 2?...Which of the following electromagnetic radiation have greater frequency?...The quantum numbers + and - for the electron spin represent :...The orbital angular momentum of an electron is $\sqrt{3}\frac{h}{\pi}$. Which of the following may be the permissible va...Electromagnetic radiations of wavelength 240 nm are just sufficient to ionize sodium atom. The ionization energy of sodi...