Atomic StructureHard
Question
In presence of HCl, metallic tin (Sn) is oxidised into stannic chloride with the help of K2Cr2O7. How many mL of 0.1 N K2Cr2O7 will be required for the oxidation of 1 gm of tin (Atomic mass of Sn = 118.7)
Options
A.226.98
B.336.98
C.405.86
D.306.98
Solution
The redox Reaction is as follows
Sn → Sn4+ + 4e-
Cr2O72- + 6e- → 2Cr3+
gmeq. of Sn = gmeq. of K2Cr2O7
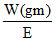 = N × V(l)
= N × V(l)
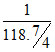 = 0.1 × V(L)
= 0.1 × V(L)
Sn → Sn4+ + 4e-
Cr2O72- + 6e- → 2Cr3+
gmeq. of Sn = gmeq. of K2Cr2O7
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In an experiment, 4g of M2Ox oxide was reduced to 2.8 g of the metal. If the atomic mass of the metal is 56 g mol-1. The...The number of unpaired electron in G. S., first E.S. and second E.S. of S (Z = 16) are, respectively,...An electron jumps from the fourth orbit to the first orbit in a H-atom. The number of photons liberated out will be...A single electron species in energy level (orbit number n) with energy X was provided with excess of energy so that it j...Quantum numbers of an atom can be defined on the basis of...