Atomic StructureHard
Question
If the value of En = - 78.4 kcal/mole, the order of the orbit in hydrogen atom is :
Options
A.4
B.3
C.2
D.1
Solution
En = - 78.4 kcal/mole = - 78.4 × 4.2 = - 329.28 kJ/mole
= -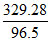 eV = - 3.4 eV. (energy of II orbit of H atom).
eV = - 3.4 eV. (energy of II orbit of H atom).
= -
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
dz2 - orbital has :...As the orbit number increases, the distance between two consecutive orbits (r1 = radius of first orbit)...The wavelength of spectral line obtained in the spectrum of ${LI}^{2 +}$ ion, when the transition takes place between tw...Radiation corresponding to the transition n = 4 to n = 2 in hydrogen atoms falls on a certain metal (work function = 2.0...There are two samples of H and He atom. Both are in some excited state. In hydrogen atom total number of lines observed ...