Atomic StructureHard
Question
Photon having energy equivalent to the binding energy of 4th state of He+ ion is used to eject an electron from the metal with K.E. 2eV. If electron is further accelerated through the potential difference of 4V then the minimum value of de-Broglie wavelength associated with the electron is :
(h = 6.6 × 10-34 J-s, me = 9.1 × 10-31 kg, 1 eV = 1.6 × 10-19J)
(h = 6.6 × 10-34 J-s, me = 9.1 × 10-31 kg, 1 eV = 1.6 × 10-19J)
Options
A.1.1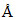
B.5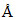
C.9.15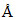
D.11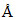
Solution
Final kinetic energy of electron, KEf = 2 + 4 = 6 eV.
∴ λ =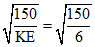 = 5
= 5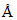
∴ λ =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Photon having energy equivalent to the binding energy of 4th state of He+ atom is used to eject an electron from the met...Incorrect statement is :-...If an electron is revolving around the nucleus of He+ ion at a distance of 4.0 Å, the magnitude of centripetal force on ...No. of visible lines when an electron returns from 5th orbit to ground state in H-spectrum :-...For which atom or ion, the energy level of the second excited state is −13.6 eV?...